Blog Archive
-
▼
2009
(338)
-
▼
October
(133)
- Eye Blindness
- Facts on Smoking
- Aids Information
- Eight Ways to get chances of success of treatment ...
- Hypertension High Blood Pressure
- Part B drug proposal
- MRI of the Breast
- Medicine Nobel Prize 2009
- Ipphone
- H1N1 Disease
- Best Diet to Lose Weight
- Is Cancer Hereditary
- Not Afraid
- Heart Disease Risk Factor
- Sickle Cell Anemia Treatment
- Natural Killer Cells
- Aids Vaccine
- Sweet play a Role in Cancer Metastasis
- Down Syndrome Information
- Effect of Carbonation
- Vulvar cancer
- Vagina Cancer
- Cancer Uterus
- Thyroid Cancer Treatment
- Cancer of the Throat
- Cancer Testicular
- Rectal Cancer
- Prostatic Cancer
- Pharyngeal Cancer
- Pancreatic Cancers
- Ovarion Cancer
- Multiple Myeloma Cancer
- Mesothelioma Information
- Melenoma
- Lymphomas
- Lung Cancers
- Liver Cancer Survival
- Penis Pimples
- Genital Herpies
- Prostitis
- Syphilis Treatment
- Nongonococcal Urethritis
- Balanitis Treatment
- Reiters Syndrome
- Sickle Cell Anemia Symptoms
- Penile Prostheses
- Peyronie's Disease Treatment
- Priapism Treatment
- Smallpox History
- Tetanus Symptoms
- Toxoplamosis
- Trichinellosis
- Tuberculosis Disease
- Rabbit Fever
- Typhoid Disease
- Chicken Pox Virus
- West Nile Disease
- Yellow Fever Virus
- Cocaine Drug
- Penis Pain Due To Related Causes Of Diabetes
- Erectile or Impotence (Penis Disease)
- Penis Rashes,Lumps & Spots
- Combat Acne with Your MP3 Player
- Skin Facts
- Stretch Marks
- What is Medical Technology
- Sexual Desire May Be Revealed in Probe of Prairie ...
- cancel the serologicaltest of hepatitis B in physi...
- MERIAL INVESTS US $ 70 MILLION
- UK Prime Minister Message Joining Charity
- Popcorn Good For You
- Breast Cancer And Health Insurance In USA
- Breast Cancer Risks
- Body Building Secrets
- American Journal of Cardiology
- Penis Disorders
- Cancer of the Penis
- Diseases of Penis
- Genetic Baldness Test
- Latest Medical Equipment
- Medical Biogel
- MRI Equipment
- hemopurifier
- Obesity Solution
- Home Made Skin Recipes
- Treatment for Oily Skin
- Leukemia Cancer
- Skin Types
- Skin Care Information
- Skin Care Tip
- Tips for Skin Care
- Beauty Skin Care Tips
- About cholera
- Infectious Viruses
- Types Infectious Diseases
- Recent Health News
- Atrial Fibrillation Guidelines
- Atrial Fibrillation Guidelines
- HIV News
- Skin Care Lines
-
▼
October
(133)
Saturday, October 3, 2009
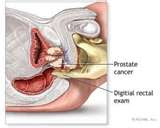
U .S . Preventive Services Task Force (USPSTF):
Screening for Prostate Cancer: U.S. Preventive Services Task Force Recommendation Statement
Description:
Update of the 2002 U.S. Preventive Services Task Force (USPSTF) recommendation statement about screening for prostate cancer.
Methods:
The USPSTF evaluated randomized, controlled trials of the benefits of prostate cancer screening; cohort and cross-sectional studies of the psychological harms of false-positive prostate-specific antigen test results; and evidence on the natural history of prostate-specific antigen–detected prostate cancer to address previously identified gaps in the evidence from the 2002 USPSTF recommendation.
Recommendations:
Current evidence is insufficient to assess the balance of benefits and harms of screening for prostate cancer in men younger than age 75 years (I statement).
Do not screen for prostate cancer in men age 75 years or older (Grade D recommendation).
The U.S. Preventive Services Task Force (USPSTF) makes recommendations about preventive care services for patients without recognized signs or symptoms of the target condition.
It bases its recommendations on a systematic review of the evidence of the benefits and harms and an assessment of the net benefit of the service.
The USPSTF recognizes that clinical or policy decisions involve more considerations than this body of evidence alone. Clinicians and policymakers should understand the evidence but individualize decision-making to the specific patient or situation.
Importance:
Prostate cancer is the most common nonskin cancer and the second leading cause of cancer death in men in the United States.
Detection:
The USPSTF found convincing evidence that prostate-specific antigen (PSA) screening can detect some cases of prostate cancer.
Benefits of Detection and Early Treatment
In men younger than age 75 years, the USPSTF found inadequate evidence to determine whether treatment for prostate cancer detected by screening improves health outcomes compared with treatment after clinical detec
ate evidence that the incremental benefits of treatment for prostate cancer detected by screening are small to none.
Harms of Detection and Early Treatment:
The USPSTF found convincing evidence that treatment for prostate cancer detected by screening causes moderate-to-substantial harms, such as erectile dysfunction, urinary incontinence, bowel dysfunction, and death. These harms are especially important because some men with prostate cancer who are treated would never have developed symptoms related to cancer during their lifetime. There is also adequate evidence that the screening process produces at least small harms, including pain and discomfort associated with prostate biopsy and psychological effects of false-positive test results.
USPSTF Assessment:
The USPSTF concludes that for men younger than age 75 years, the benefits of screening for prostate cancer are uncertain and the balance of benefits and harms cannot be determined.
For men 75 years or older, there is moderate certainty that the harms of screening for prostate cancer outweigh the benefits.
Clinical Considerations
Patient Population under Consideration:
This recommendation applies to men in the general U.S. population.
Risk Assessment:
Older men, African-American men, and men with a family history of prostate cancer are at increased risk for diagnosis of and death from prostate cancer (1). Unfortunately, the previously described gaps in the evidence regarding potential benefits of screening also apply to these men.
Screening Tests:
The PSA test is more sensitive than the digital rectal examination for detecting prostate cancer. The conventional PSA screening cut-point of 4.0 µg/L detects many cases of prostate cancer; however, some early cases will be missed by this cut-point (2, 3). Using a lower cut-point to define an abnormal PSA level detects more cases of cancer.
The proportion of cancer cases detected by lower cut-points that would ever become clinically apparent is unknown; lower cut-points would label many more men as potentially having cancer. For example, lowering the PSA cut-point to 2.5 µg/L would more than double the number of U.S. men between 40 and 69 years of age with abnormal results (4).
Variations of PSA screening, including the use of age-adjusted PSA cut-points, free PSA, PSA density, PSA velocity, PSA slope, and PSA doubling time, have been proposed to improve detection of "clinically important" prostate cancer cases. However, no evidence suggests that any of these testing strategies improves health outcomes (2, 5).
Suggestions for Practice:
Given the uncertainties and controversy surrounding prostate cancer screening in men younger than age 75 years, a clinician should not order the PSA test without first discussing with the patient the potential but uncertain benefits and the known harms of prostate cancer screening and treatment. Men should be informed of the gaps in the evidence and should be assisted in considering their personal preferences before deciding whether to be tested.
Treatment:
Because of the uncertainty about the benefits of treating prostate cancer detected by screening men younger than age 75 years, there is no consensus regarding optimal treatment. Current management strategies for localized prostate cancer include watchful waiting (observation with palliative treatment for symptoms only), active surveillance (periodic biochemical monitoring with conversion to curative treatment for signs of disease progression), radical prostatectomy, external-beam radiation therapy, and brachytherapy (or radioactive seed implantation therapy) (6).
If treatment for prostate cancer detected by screening improves health outcomes, the population most likely to benefit from screening will be men age 50 to 74 years. Even if prostate cancer screening is determined to be effective, the length of time required to experience a mortality benefit is greater than 10 years. Because a 75-year-old man has an average life expectancy of about 10 years, very few men age 75 years or older would experience a mortality benefit. Similarly, men younger than age 75 years who have chronic medical problems and a life expectancy of fewer than 10 years are also unlikely to benefit from screening and treatment (2).
Screening Intervals:
The yield of screening in terms of cancer cases detected declines rapidly with repeated annual testing. If screening were to reduce deaths, PSA screening as infrequently as every 4 years could yield as much of a benefit as annual screening (7).
Useful Resources
Shared decision-making resources specific to prostate cancer screening for clinicians and patients are available from the Centers of Disease Control and Prevention (http://www.cdc.gov/cancer/prostate/publications/).
Other Considerations:
Research Needs and Gaps
Good-quality randomized, controlled trials (RCTs) are needed to establish the effect, if any, of population-based PSA screening on prostate cancer mortality in men younger than age 75 years. The results of 2 ongoing trials, the U.S. Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial and the European Study of Screening for Prostate Cancer, should help to clarify the potential benefits of screening.
Future studies should identify testable characteristics of screening-detected prostate cancer that reliably predict poor health outcomes and that therefore may be indications for treatment. Research is needed to compare the long-term benefits of immediate treatment with delayed treatment in men with screening-detected prostate cancer. Two ongoing RCTs, the U.S. Prostate Intervention Versus Observation Trial and the U.K. Prostate Testing for Cancer and Treatment Study, are studying these issues.
Discussion
Burden of Disease
An estimated 218 890 U.S. men received a prostate cancer diagnosis in 2007, and 1 of 6 men in the U.S. will receive the diagnosis in his lifetime (8). An estimated 27 350 men died of prostate cancer in the United States in 2006 (1). The median age of death from prostate cancer from 2000 through 2004 was 80 years, and 71% of deaths occurred in men older than 75 years. African-American men have a substantially higher prostate cancer incidence rate than white men (217.5 vs. 134.5 cases per 100 000 men) and more than twice the prostate cancer mortality rate of white men (56.1 vs. 23.4 deaths per 100 000 men) (9).
Prostate cancer is a clinically heterogeneous disease. A substantial proportion of prostate cancer cases detected with current screening methods will never cause symptoms during the patients' lifetime. Modeling studies based on U.S. incidence data suggest overdiagnosis rates ranging from 29% to 44% of all prostate cancer cases detected by PSA screening (10). Because patients with "pseudo-disease" receive no benefit from, and may be harmed by, prostate cancer screening and treatment, prostate cancer detection in this population constitutes an important burden.
Scope of USPSTF Review
The previous review, performed for the USPSTF in 2002, found insufficient evidence that screening for prostate cancer improved health outcomes, including mortality. It also found little evidence on the harms of the screening process or the natural history of prostate cancer cases detected with screening (2). The USPSTF determined that a focused evidence update (5) should systematically review direct evidence that PSA screening reduces morbidity and mortality, evidence on the magnitude and nature of harms associated with false-positive screening results, and evidence on health outcomes of patients with screening-detected prostate cancer who did not receive active treatment.
Accuracy of Screening Tests
The 2002 review noted inherent problems with the use of needle biopsy results as a reference standard to assess the accuracy of prostate cancer screening tests. Biopsy detection rates vary according to the number of biopsies performed during a single procedure: The more biopsies performed, the more cancer cases detected. More cancer cases detected with a "saturation" (≥20) biopsy procedure tend to increase the apparent specificity of an elevated PSA level; however, many additional cancer cases detected this way are likely to be clinically unimportant. Thus, the accuracy of the PSA test for detecting clinically important prostate cancer cases cannot be determined with precision.
Longitudinal follow-up has also been used as a reference standard. A retrospective study found the sensitivity of a PSA level of 4.0 µg/L or higher to be about 91% for detecting aggressive cases of prostate cancer that developed within 2 years of screening; the sensitivity was about 56% for detecting nonaggressive cancer cases within the same period. Among men who did not receive a prostate cancer diagnosis within 10 years, 9% had an initial PSA level of 4.0 µg/L or greater (which translates to a specificity of 91% for any prostate cancer) (11).
Effectiveness of Early Detection and Treatment
A meta-analysis of 2 poor-quality RCTs of population-based screening for prostate cancer using PSA and digital rectal examination found no reduction in prostate cancer mortality in men invited versus men not invited for screening (relative risk, 1.01 [95% CI, 0.80 to 1.29]) (12). A recent RCT reported that men who received PSA screening had a decreased risk for receiving a diagnosis of metastatic prostate cancer (13). The USPSTF assessed the study as providing inconclusive evidence of benefit from screening because of a high likelihood of unequal outcome ascertainment and small absolute numbers of an imperfect intermediate health outcome (metastatic prostate cancer is an imperfect surrogate of prostate cancer mortality because of both high initial response rates to androgen deprivation therapy and competing causes of death). No RCTs have reported health outcomes from the variations of PSA screening that consist of multiple measurements over time (for example, measurements of PSA velocity, PSA slope, or PSA doubling time).
Randomized, controlled trials comparing prostate cancer treatments with watchful waiting have enrolled few patients with screening-detected prostate cancer. An RCT of 695 men with localized prostate cancer reported a small absolute reduction in all-cause mortality in patients assigned to radical prostatectomy; however, only 5.2% of participants had screening-detected prostate cancer, more than 40% presented with symptoms, and 77.8% of the treatment group had stage T2 (palpable) cancer (14). This stage of cancer is more advanced than cancer typically detected by screening. Yet, after a median of 8.2 years, only 14.4% of men in the control group and 8.6% of men in the treatment group had died of prostate cancer.
Screening-detected cancer is biologically less aggressive, is being detected much earlier in its natural history, or both, so it is unlikely that these results could be obtained in a study of screening-detected cancer in this same time frame. Even if the same disease-specific results could be obtained with a longer time frame, competing causes of death would make any reduction in all-cause mortality less than that found in the study. It is noteworthy that in the 372 men who were at least 65 years of age at the time of diagnosis, the 10-year incidence of death from prostate cancer was similar between the watchful waiting and radical prostatectomy groups, suggesting no benefit from surgery in this age group (14).
Estimate of Magnitude of Net Benefit
In men younger than age 75 years, the USPSTF could not determine the net benefit of screening for prostate cancer because of low certainty about the magnitude of benefits of screening and treatment.
In men age 75 years or older, the USPSTF found no direct evidence of benefits of prostate cancer screening. However, the USPSTF was able to establish an upper bound for the potential magnitude of the benefit of treating screening-detected prostate cancer in this age group, by extrapolating from evidence of treatment for clinically detected prostate cancer in this age group (14). For a population of men with an average life expectancy of 10 years or fewer, the USPSTF determined that the benefits of prostate cancer screening and treatment would range from small to none.
Weighing this magnitude of benefit against the moderate-to-substantial psychological and physical harms associated with prostate cancer screening and treatment, the USPSTF concluded that there is at least moderate certainty that the harms of screening for prostate cancer in men age 75 years or older outweigh the benefits.
How Does Evidence Fit with Biological Understanding?
Prostate-specific antigen screening presupposes that most asymptomatic prostate cancer cases will ultimately become symptomatic cases that lead to poor health outcomes. However, the natural history of PSA-detected, nonpalpable, localized prostate cancer is poorly described. No prospective studies have followed a population-based cohort of patients with screening-detected cancer who have had no intervention in order to determine health outcomes resulting from natural progression of the disease. Evidence from small, selected cohorts of men with arbitrarily defined "favorable risk" (that is, with prostate cancer likely to be clinically indolent) suggest a good prognosis for some men with screening-detected cancer; however, the longest of these studies has reported health outcomes from 2 to 10 years after diagnosis only (5).
Update of Previous USPSTF Recommendation
This recommendation replaces the 2002 recommendation. The major change in the current recommendation is that the USPSTF now recommends against screening men age 75 years or older for prostate cancer.
Recommendations of Other Groups
Most major U.S. medical organizations recommend that clinicians discuss the potential benefits and known harms of PSA screening with their patients, consider their patients' preferences, and individualize screening decisions. They generally agree that the most appropriate candidates for screening include men age 50 years or older who have a life expectancy of at least 10 years. These organizations include the American Academy of Family Physicians (15), American College of Physicians (16), American College of Preventive Medicine (17), and American Medical Association. The American Cancer Society (18) and American Urological Association (19) recommend offering PSA measurement and digital rectal examination to men annually beginning at age 50 years.
Labels: Cancer Forum
0 Comments:
Category
- Breast cancer (1)
- Cancer Forum (46)
- Depression and Exercise (17)
- Fitness Woman (14)
- Health Video (22)
- herbal remedies (63)
- Infectious Deseases (14)
- joint pain exercises (8)
- Latest Medical Technology (4)
- loose fat exercise (13)
- Penis Diseases (17)
- Pregnancy (6)
- Pregnancy Exercise (27)
- pregnancy tips video (21)
- skin care (15)
- World Health News (62)